Our Research
Our Research
Research
The Caradonna Laboratory is interested in studies involving airway progenitor cell differentiation and airway mucociliary tissue homeostasis. We have adopted an air-liquid interface model system to grow and differentiate airway cells derived from the nasal passages of donors. Culturing primary human airway progenitor cells at the air-liquid interface triggers their differentiation and the progenitor cells will regenerate fully functioning airway mucociliary tissue. The formation of the tissues in culture closely approximates tissue injury repair pathways that occur in the human body and the fully differentiated mucociliary tissue cultures can be maintained for months in culture. This model system of airway mucociliary tissue regeneration and homeostasis is useful for studies aimed at understanding how drugs, vitamins, supplements, and environmental toxins impact these pathways. Our laboratory also utilizes a range of cell-lines for studies in protein localization, protein-protein interactions, and transcriptional assays. Some techniques we commonly use include PCR, qPCR, site-directed mutagenesis, transfection, transduction, western blotting, immunoprecipitation, dual-luciferase assays, immunocytochemistry, immunohistochemistry, and flow cytometry.
Airway mucociliary tissue model system
We isolate primary airway progenitor cells from the nasal turbinates of donors and use the cells to grow airway mucociliary tissue in culture.
Our laboratory is designed to use a human-derived, in-vitro model of airway mucociliary tissue. We isolate Human Nasal Epithelial Cells (HNECs) from the nasal mucociliary tissue of donors with a small brush and expand the airway stem cells in culture. The HNECs are then seeded onto semi-permeable transwell inserts that are in contact with the culture media in the lower chamber but don’t have media in the upper chamber where the cells adhere. This allows the cells to be exposed to the air and this is known as the Air-Liquid Interface (ALI). HNECs cultured at the ALI initiate a differentiation program and the cells regenerate fully functioning airway mucociliary tissue that maintains the morphology and cell types of mucociliary tissue located in the lung, bronchus, and nasal passages. Fully differentiated ALI-tissue can be maintained in culture for weeks at a time for long-term experiments. Tissues can be harvested for protein, DNA, or RNA, or can be embedded in paraffin wax and sectioned for histological analyses.
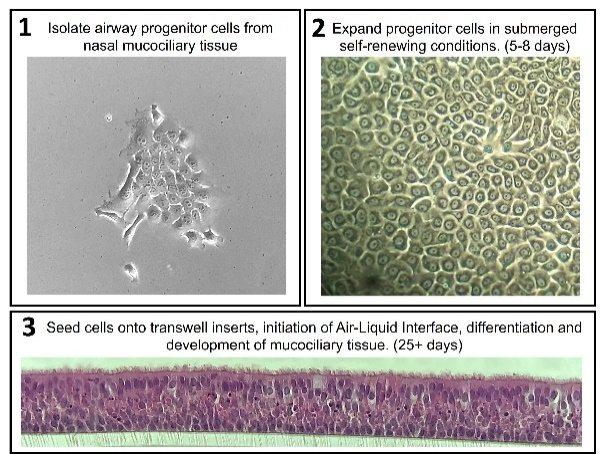 Figure 1: Schematic of airway mucociliary tissue culture model system. Airway progenitor cells are isolated by gently brushing the nasal passages of donors and expanded in culture. Once there are enough progenitor cells, they can be seeded onto transwell inserts for air-liquid interface culture and differentiation into mucociliary tissue.
Figure 1: Schematic of airway mucociliary tissue culture model system. Airway progenitor cells are isolated by gently brushing the nasal passages of donors and expanded in culture. Once there are enough progenitor cells, they can be seeded onto transwell inserts for air-liquid interface culture and differentiation into mucociliary tissue.
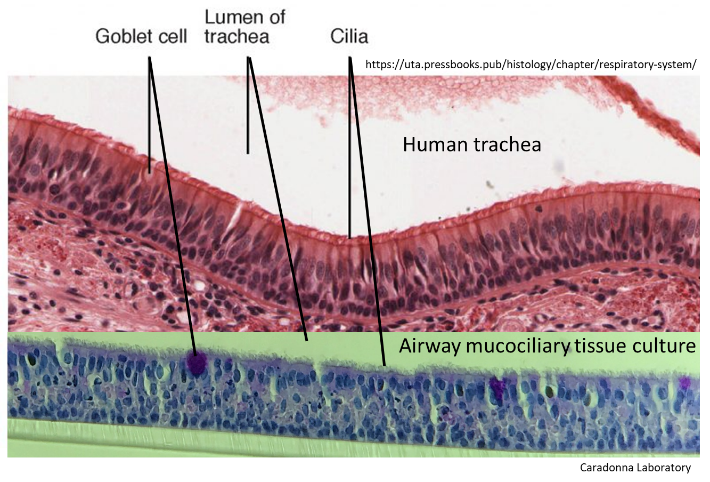 Figure 2: Comparison of human tracheal tissue structure (Top) to the structure of our human airway mucociliary tissue cultures generated from nasal progenitor cells (Bottom).
Figure 2: Comparison of human tracheal tissue structure (Top) to the structure of our human airway mucociliary tissue cultures generated from nasal progenitor cells (Bottom).
The Effects of E-cigarette Vapors on airway mucociliary tissue
Exploring how chronic exposure to e-cigarette vapors alters the function and structure of airway mucociliary tissue.
Electronic cigarettes (e-cigarettes) and vape-pens are handheld battery-powered devices that contain a vape-liquid which is used to produce a vapor that is inhaled by the user. The active ingredients in commercial vape-liquids are commonly nicotine, tetrahydrocannabinol or cannabidiol. The prevalent use of e-cigarettes by both adult and young adult populations and the 2019 emergence of an e-cigarette related illness has made understanding how e-cigarette vapors affect our respiratory tissues a public health concern. Our laboratory has taken advantage of 3D-Printing and microcontroller technology to develop a compact automated e-cigarette research device capable of operating e-cigarettes and delivering the vapor to a multi-well cell culture plate. We have combined our human airway mucociliary tissue model system with our vaping device for investigations in airway tissue consequences from chronic e-cigarette usage. By using primary human airway tissue cultures, we can focus on identifying cellular responses and consequences specific to airway mucociliary tissue, removing crosstalk or interference from immune cell or hormonal responses which may occur in animal-based model systems. A more systematic analysis of the consequences to the airway when chronically vaping will aide in navigating the future safety measures for e-cigarette users.
 Figure 3: Laboratory designed e-cigarette research device 3D-printed from polylactic acid plastic and controlled with an Arduino microcontroller. The device can draw vapor from e-cigarettes and deposit the vapor to an exposure chamber containing human airway mucociliary tissue cultures.
Figure 3: Laboratory designed e-cigarette research device 3D-printed from polylactic acid plastic and controlled with an Arduino microcontroller. The device can draw vapor from e-cigarettes and deposit the vapor to an exposure chamber containing human airway mucociliary tissue cultures.
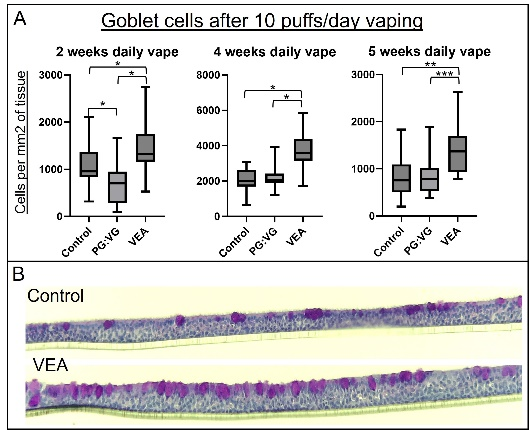 Figure 4: Airway tissue cultures chronically exposed to vapors generated from vitamin E acetate (VEA) contained a greater average amount of goblet cells compared to tissues exposed to propylene glycol/vegetable glycerin vapors, or control tissues.
Figure 4: Airway tissue cultures chronically exposed to vapors generated from vitamin E acetate (VEA) contained a greater average amount of goblet cells compared to tissues exposed to propylene glycol/vegetable glycerin vapors, or control tissues.
We would like to acknowledge the generous support of the New Jersey Health Foundation, which provided funds for this research (NJHF Grant PC68-21).
Respiratory ACE2
Using human mucociliary airway tissue cultures to model the production of exosome associated ACE2 in respiratory mucous.
In the early stages of the COVID-19 pandemic Dr. Caradonna and I asked ourselves what we could do to assist in the research effort. Since the main route of entry for SARS-CoV-2 particles is through airway mucociliary tissue, and our laboratory utilizes this tissue as a model system, we decided to focus our research endeavor on the cellular receptor necessary for viral entry, angiotensin converting enzyme 2 (ACE2). We demonstrated that human airway mucociliary tissue expresses three distinct ACE2 transcript variants, which encode for protein that vary in their extreme c-termini which may infer isoform-specific functions. We used fluorescent immunohistochemistry to establish that ACE2 protein localization in airway mucociliary tissue will vary depending on the antibody used. These findings suggest the existence of ACE2 protein-protein interactions or modifications specific to cellular localization. We have also observed that our airway mucociliary tissue cultures mimic in-vivo respiratory tissue by shedding ACE2 into respiratory mucous both as a soluble cleaved ACE2 and as an exosome-associated full-length ACE2. We are interested in using our model system to investigate molecular pathways which may modulate the balance of soluble versus exosome-associated ACE2, and how the mucosal concentrations of these proteins affect susceptibility to SARS-CoV-2 infection.
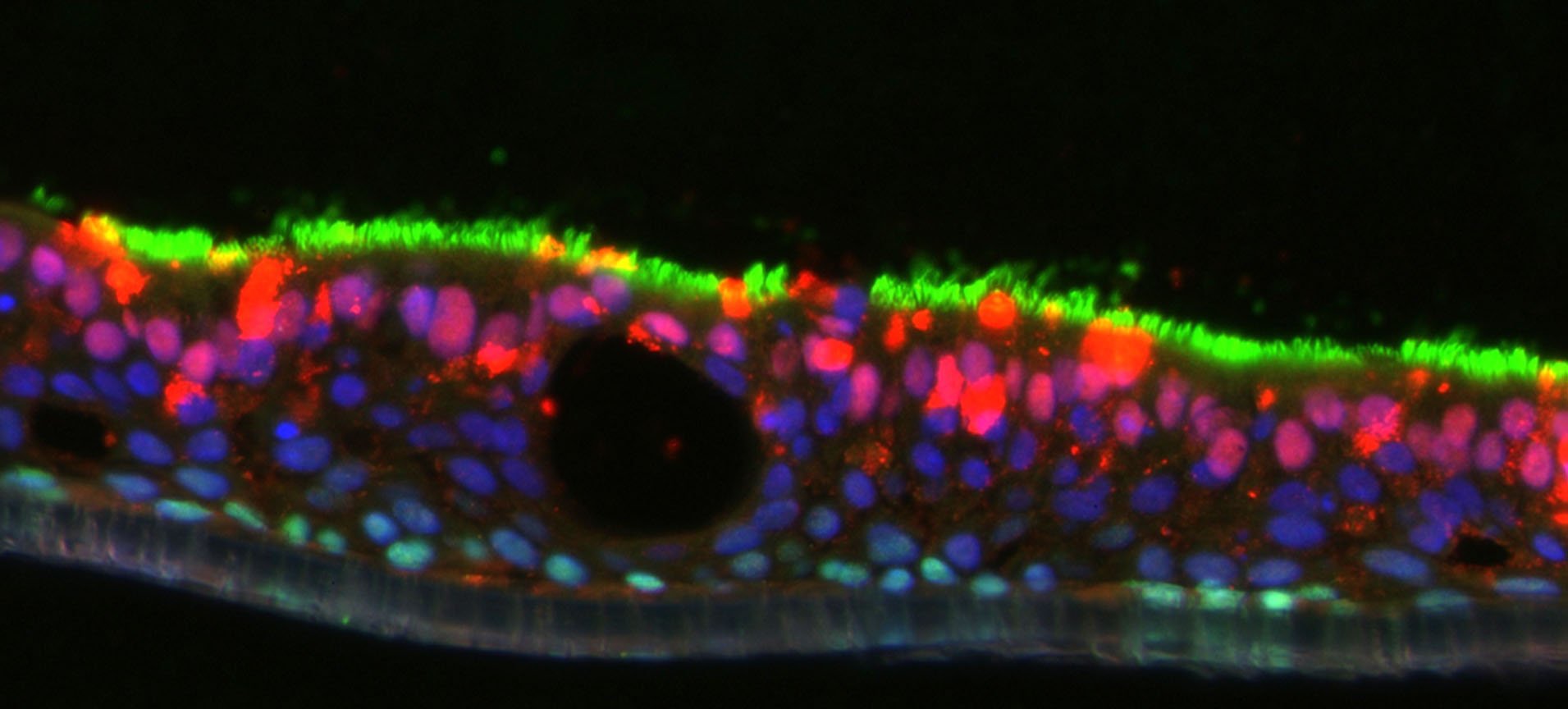
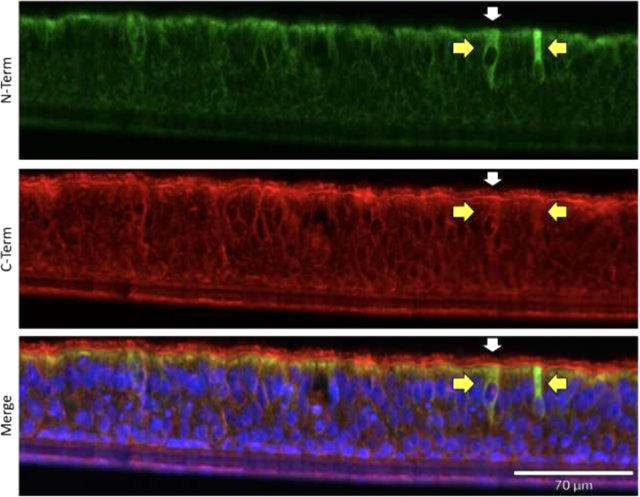 Figure 5: ACE2 localization utilizing fluorescent immunohistochemistry. Two different antibodies, one to the N-terminus and the other to the C-terminal half of the protein were chosen. Although both antibodies displayed the same signals when used in our western blot analysis, they display distinct and overlapping signals via fluorescent immunohistochemistry. The apical cytosol of ciliated cells contained a mixture of individual signals and overlapping signals from both ACE2 antibodies (Yellow arrows highlight example of strong N-term Ab signal with minimal C-term Ab signal.) Only the ACE2 C-term antibody displayed plasma membrane signal on ciliated cells (White arrow highlights an example of C-term Ab membrane localization). Bottom panel -merged image including DAPI staining of nuclei.
Figure 5: ACE2 localization utilizing fluorescent immunohistochemistry. Two different antibodies, one to the N-terminus and the other to the C-terminal half of the protein were chosen. Although both antibodies displayed the same signals when used in our western blot analysis, they display distinct and overlapping signals via fluorescent immunohistochemistry. The apical cytosol of ciliated cells contained a mixture of individual signals and overlapping signals from both ACE2 antibodies (Yellow arrows highlight example of strong N-term Ab signal with minimal C-term Ab signal.) Only the ACE2 C-term antibody displayed plasma membrane signal on ciliated cells (White arrow highlights an example of C-term Ab membrane localization). Bottom panel -merged image including DAPI staining of nuclei.
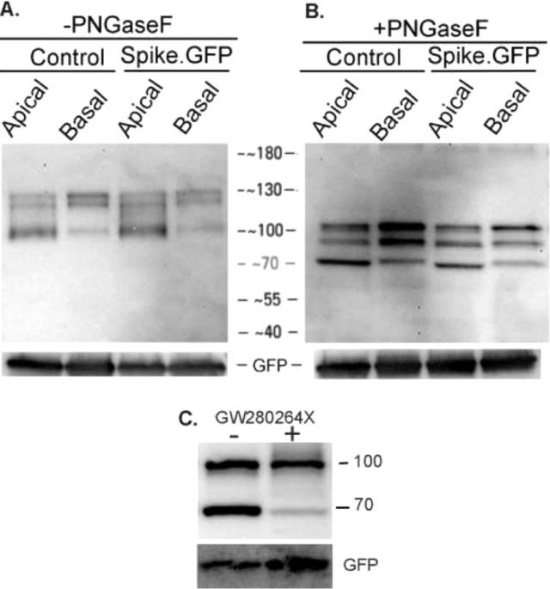 Figure 6: Apical and basal nature of ACE2. 25-day ALI cultures were exposed to 20 µg of spike-GFP fusion protein, either apically or at the basal surface for 3 h at 37C in a 5% CO 2 incubator. At the end of the incubation samples were harvested, centrifuged and subjected to affinity pull-down. Control indicates PBS alone (apical) or media alone (basal). Control PBS and media were harvested, centrifuged and then 20 µg of spike-GFP protein was added and ACE2 was then affinity isolated. (A.) As indicated in the figure it appears that ACE2 is released in the absence of any inducing agent. Equivalent amounts of ACE2 are seen in the absence or presence of spike-GFP protein. (B.) Affinity isolated samples were treated with PNGase F. There are slight differences in ACE2 gel migration between isolates derived from the apical and basal surfaces. (C.) ALI cultures were incubated without or with 20 µM GW280264X, a dual ADAM10 and ADAM17 protease inhibitor for 14 h on the apical surface and processed as described above. As seen in the figure, the protease inhibitor decreases the amount of the 70 kg dalton band. This is indicative of protease-dependent shedding of the ACE2 protein.
Figure 6: Apical and basal nature of ACE2. 25-day ALI cultures were exposed to 20 µg of spike-GFP fusion protein, either apically or at the basal surface for 3 h at 37C in a 5% CO 2 incubator. At the end of the incubation samples were harvested, centrifuged and subjected to affinity pull-down. Control indicates PBS alone (apical) or media alone (basal). Control PBS and media were harvested, centrifuged and then 20 µg of spike-GFP protein was added and ACE2 was then affinity isolated. (A.) As indicated in the figure it appears that ACE2 is released in the absence of any inducing agent. Equivalent amounts of ACE2 are seen in the absence or presence of spike-GFP protein. (B.) Affinity isolated samples were treated with PNGase F. There are slight differences in ACE2 gel migration between isolates derived from the apical and basal surfaces. (C.) ALI cultures were incubated without or with 20 µM GW280264X, a dual ADAM10 and ADAM17 protease inhibitor for 14 h on the apical surface and processed as described above. As seen in the figure, the protease inhibitor decreases the amount of the 70 kg dalton band. This is indicative of protease-dependent shedding of the ACE2 protein.